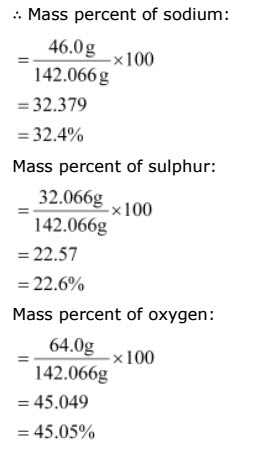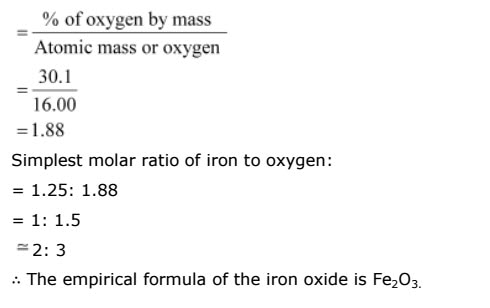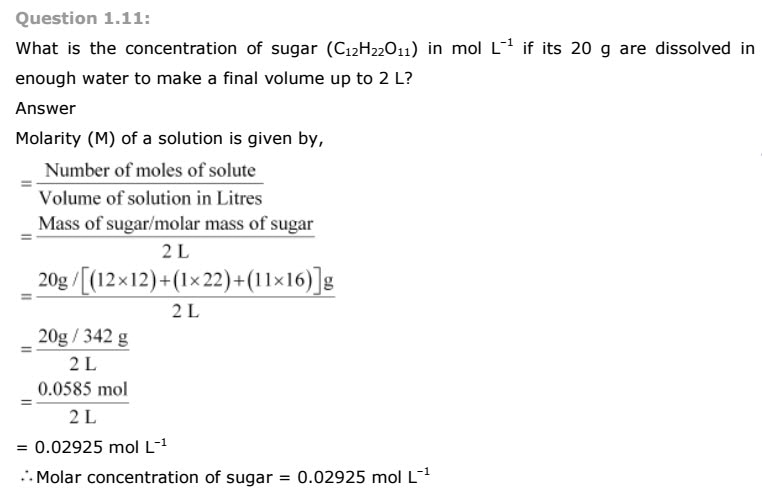SOME BASIC CONCEPTS OF CHEMISTRY
-
1.1
-
1.2
-
1.3
-
1.4
-
1.5
-
1.6
-
1.7
-
1.8
-
1.9
-
1.10
<
>
Question-Calculate the molecular mass of the following: (i) H2O (ii) CO2 (iii) CH4
ANSWER:-
(i) H2O:
The molecular mass of water, H2O
= (2 × Atomic mass of hydrogen) + (1 × Atomic mass of oxygen)
= [2(1.0084) + 1(16.00 u)]
= 2.016 u + 16.00 u
= 18.016
= 18.02 u
(ii) CO2:
The molecular mass of carbon dioxide, CO2
= (1 × Atomic mass of carbon) + (2 × Atomic mass of oxygen)
= [1(12.011 u) + 2 (16.00 u)]
= 12.011 u + 32.00 u
= 44.01 u
(iii) CH4:
The molecular mass of methane, CH4
= (1 × Atomic mass of carbon) + (4 × Atomic mass of hydrogen)
= [1(12.011 u) + 4 (1.008 u)]
= 12.011 u + 4.032 u
= 16.043 u
Question 1.2:
Calculate the mass percent of different elements present in sodium sulphate (Na2SO4).
Answer
The molecular formula of sodium sulphate is Na2So4
Molar mass of Na2SO4= [(2 × 23.0) + (32.066) + 4 (16.00)]
= 142.066 g
Mass percent of an element= Mass of Element in the compound / molar mass of the compound x 100
Calculate the mass percent of different elements present in sodium sulphate (Na2SO4).
Answer
The molecular formula of sodium sulphate is Na2So4
Molar mass of Na2SO4= [(2 × 23.0) + (32.066) + 4 (16.00)]
= 142.066 g
Mass percent of an element= Mass of Element in the compound / molar mass of the compound x 100
Question 1.4:
Calculate the amount of carbon dioxide that could be produced when
(i) 1 mole of carbon is burnt in air.
(ii) 1 mole of carbon is burnt in 16 g of dioxygen.
(iii) 2 moles of carbon are burnt in 16 g of dioxygen.
Answer
The balanced reaction of combustion of carbon can be written as:
(i) As per the balanced equation, 1 mole of carbon burns in1 mole of dioxygen (air) to
produce1 mole of carbon dioxide.
(ii) According to the question, only 16 g of dioxygen is available. Hence, it will react with
0.5 mole of carbon to give 22 g of carbon dioxide. Hence, it is a limiting reactant.
(iii) According to the question, only 16 g of dioxygen is available. It is a limiting
reactant. Thus, 16 g of dioxygen can combine with only 0.5 mole of carbon to give 22 g
of carbon dioxide.
Calculate the amount of carbon dioxide that could be produced when
(i) 1 mole of carbon is burnt in air.
(ii) 1 mole of carbon is burnt in 16 g of dioxygen.
(iii) 2 moles of carbon are burnt in 16 g of dioxygen.
Answer
The balanced reaction of combustion of carbon can be written as:
(i) As per the balanced equation, 1 mole of carbon burns in1 mole of dioxygen (air) to
produce1 mole of carbon dioxide.
(ii) According to the question, only 16 g of dioxygen is available. Hence, it will react with
0.5 mole of carbon to give 22 g of carbon dioxide. Hence, it is a limiting reactant.
(iii) According to the question, only 16 g of dioxygen is available. It is a limiting
reactant. Thus, 16 g of dioxygen can combine with only 0.5 mole of carbon to give 22 g
of carbon dioxide.
Question 1.5:
Calculate the mass of sodium acetate (CH3COONa) required to make 500 mL of 0.375
molar aqueous solution. Molar mass of sodium acetate is 82.0245 g mol–1
Answer
0.375 M aqueous solution of sodium acetate
≡ 1000 mL of solution containing 0.375 moles of sodium acetate
∴Number of moles of sodium acetate in 500 mL
=0.375/1000 x 500
=0.1875 mole
Molar mass of sodium acetate = 82.0245 g mole–1 (Given)
∴ Required mass of sodium acetate = (82.0245 g mol–1) (0.1875 mole)
= 15.38 g
Calculate the mass of sodium acetate (CH3COONa) required to make 500 mL of 0.375
molar aqueous solution. Molar mass of sodium acetate is 82.0245 g mol–1
Answer
0.375 M aqueous solution of sodium acetate
≡ 1000 mL of solution containing 0.375 moles of sodium acetate
∴Number of moles of sodium acetate in 500 mL
=0.375/1000 x 500
=0.1875 mole
Molar mass of sodium acetate = 82.0245 g mole–1 (Given)
∴ Required mass of sodium acetate = (82.0245 g mol–1) (0.1875 mole)
= 15.38 g
Calculate the concentration of nitric acid in moles per litre in a sample which has a
density, 1.41 g mL–1 and the mass per cent of nitric acid in it being 69%.
Answer
Mass percent of nitric acid in the sample = 69 % [Given]
Thus, 100 g of nitric acid contains 69 g of nitric acid by mass.
Molar mass of nitric acid (HNO3)
= {1 + 14 + 3(16)} g mol–1
= 1 + 14 + 48
= 63 g mol–1
∴ Number of moles in 69 g of HNO3
density, 1.41 g mL–1 and the mass per cent of nitric acid in it being 69%.
Answer
Mass percent of nitric acid in the sample = 69 % [Given]
Thus, 100 g of nitric acid contains 69 g of nitric acid by mass.
Molar mass of nitric acid (HNO3)
= {1 + 14 + 3(16)} g mol–1
= 1 + 14 + 48
= 63 g mol–1
∴ Number of moles in 69 g of HNO3
Question 1.7:
How much copper can be obtained from 100 g of copper sulphate (CuSO4)?
Answer
1 mole of CuSO4 contains 1 mole of copper.
Molar mass of CuSO4 = (63.5) + (32.00) + 4(16.00)
= 63.5 + 32.00 + 64.00
= 159.5 g
159.5 g of CuSO4 contains 63.5 g of copper.
⇒ 100 g of CuSO4 will contain 63.5 x 100g / 159.5 of copper.
Amount of copper that can be obtained from 100 g CuSO4 63.5 x 100g / 159.5
= 39.81 g
How much copper can be obtained from 100 g of copper sulphate (CuSO4)?
Answer
1 mole of CuSO4 contains 1 mole of copper.
Molar mass of CuSO4 = (63.5) + (32.00) + 4(16.00)
= 63.5 + 32.00 + 64.00
= 159.5 g
159.5 g of CuSO4 contains 63.5 g of copper.
⇒ 100 g of CuSO4 will contain 63.5 x 100g / 159.5 of copper.
Amount of copper that can be obtained from 100 g CuSO4 63.5 x 100g / 159.5
= 39.81 g
Question 1.8:
Determine the molecular formula of an oxide of iron in which the mass per cent of iron
and oxygen are 69.9 and 30.1 respectively. Given that the molar mass of the oxide is
159.69 g mol–1.
Answer
Mass percent of iron (Fe) = 69.9% (Given)
Mass percent of oxygen (O) = 30.1% (Given)
Number of moles of iron present in the oxide =69.90/55.85
= 1.25
Number of moles of oxygen present in the oxide =30.1/16.0
= 1.88
Ratio of iron to oxygen in the oxide,
1.25 :1.88
= 1 : 1.5
= 2 : 3
The empirical formula of the oxide is Fe2O3.
Empirical formula mass of Fe2O3 = [2(55.85) + 3(16.00)] g
Molar mass of Fe2O3 = 159.69 g
Molecular formula of a compound is obtained by multiplying the empirical formula with n.
Thus, the empirical formula of the given oxide is Fe2O3 and n is 1.
Hence, the molecular formula of the oxide is Fe2O3.
Determine the molecular formula of an oxide of iron in which the mass per cent of iron
and oxygen are 69.9 and 30.1 respectively. Given that the molar mass of the oxide is
159.69 g mol–1.
Answer
Mass percent of iron (Fe) = 69.9% (Given)
Mass percent of oxygen (O) = 30.1% (Given)
Number of moles of iron present in the oxide =69.90/55.85
= 1.25
Number of moles of oxygen present in the oxide =30.1/16.0
= 1.88
Ratio of iron to oxygen in the oxide,
1.25 :1.88
= 1 : 1.5
= 2 : 3
The empirical formula of the oxide is Fe2O3.
Empirical formula mass of Fe2O3 = [2(55.85) + 3(16.00)] g
Molar mass of Fe2O3 = 159.69 g
Molecular formula of a compound is obtained by multiplying the empirical formula with n.
Thus, the empirical formula of the given oxide is Fe2O3 and n is 1.
Hence, the molecular formula of the oxide is Fe2O3.
-
1.11
-
1.12
-
1.13
-
1.14
-
1.15
-
1.16
-
1.17
-
1.18
-
1.19
-
1.20
<
>
Question 1.13:
Pressure is determined as force per unit area of the surface. The SI unit of pressure,
Pascal is as shown below:
1Pa = 1N m–2
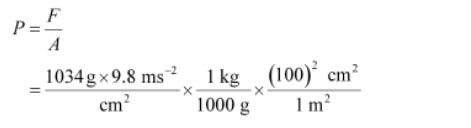
If mass of air at sea level is 1034 g cm–2, calculate the pressure in Pascal.
Answer
Pressure is defined as force acting per unit area of the surface.
Pressure is determined as force per unit area of the surface. The SI unit of pressure,
Pascal is as shown below:
1Pa = 1N m–2
If mass of air at sea level is 1034 g cm–2, calculate the pressure in Pascal.
Answer
Pressure is defined as force acting per unit area of the surface.
We know,
1 N = 1 kg ms–2
Then,
1 Pa = 1 Nm–2 = 1 kg m–2s^–2
1 Pa = 1 kg m–1s^–2
Pressure = 1.01332 × 10^5 Pa
1 N = 1 kg ms–2
Then,
1 Pa = 1 Nm–2 = 1 kg m–2s^–2
1 Pa = 1 kg m–1s^–2
Pressure = 1.01332 × 10^5 Pa
Question 1.14:
What is the SI unit of mass? How is it defined?
Answer
The SI unit of mass is kilogram (kg). 1 Kilogram is defined as the mass equal to the
mass of the international prototype of kilogram.
What is the SI unit of mass? How is it defined?
Answer
The SI unit of mass is kilogram (kg). 1 Kilogram is defined as the mass equal to the
mass of the international prototype of kilogram.
Question 1.16:
What do you mean by significant figures?
Answer
Significant figures are those meaningful digits that are known with certainty.
They indicate uncertainty in an experiment or calculated value. For example, if 15.6 mL
is the result of an experiment, then 15 is certain while 6 is uncertain, and the total
number of significant figures are 3.
Hence, significant figures are defined as the total number of digits in a number including
the last digit that represents the uncertainty of the result.
What do you mean by significant figures?
Answer
Significant figures are those meaningful digits that are known with certainty.
They indicate uncertainty in an experiment or calculated value. For example, if 15.6 mL
is the result of an experiment, then 15 is certain while 6 is uncertain, and the total
number of significant figures are 3.
Hence, significant figures are defined as the total number of digits in a number including
the last digit that represents the uncertainty of the result.
Question 1.19:
How many significant figures are present in the following?
Answers
(i) 0.0025
(ii) 208
(iii) 5005
(iv) 126,000
(v) 500.0
(vi) 2.0034
Answer
(i) 0.0025
There are 2 significant figures.
(ii) 208
There are 3 significant figures.
(iii) 5005
There are 4 significant figures.
(iv) 126,000
There are 3 significant figures.
(v) 500.0
There are 4 significant figures.
(vi) 2.0034
There are 5 significant figures.
How many significant figures are present in the following?
Answers
(i) 0.0025
(ii) 208
(iii) 5005
(iv) 126,000
(v) 500.0
(vi) 2.0034
Answer
(i) 0.0025
There are 2 significant figures.
(ii) 208
There are 3 significant figures.
(iii) 5005
There are 4 significant figures.
(iv) 126,000
There are 3 significant figures.
(v) 500.0
There are 4 significant figures.
(vi) 2.0034
There are 5 significant figures.
Question 1.20:
Round up the following upto three significant figures:
(i) 34.216
(ii) 10.4107
(iii) 0.04597
(iv) 2808
Answer
(i) 34.2
(ii) 10.4
(iii) 0.0460
(iv) 2810
Round up the following upto three significant figures:
(i) 34.216
(ii) 10.4107
(iii) 0.04597
(iv) 2808
Answer
(i) 34.2
(ii) 10.4
(iii) 0.0460
(iv) 2810
Next 16 questions coming soon we are working on it